What is the Global Lateral Flow Assays Market Size?
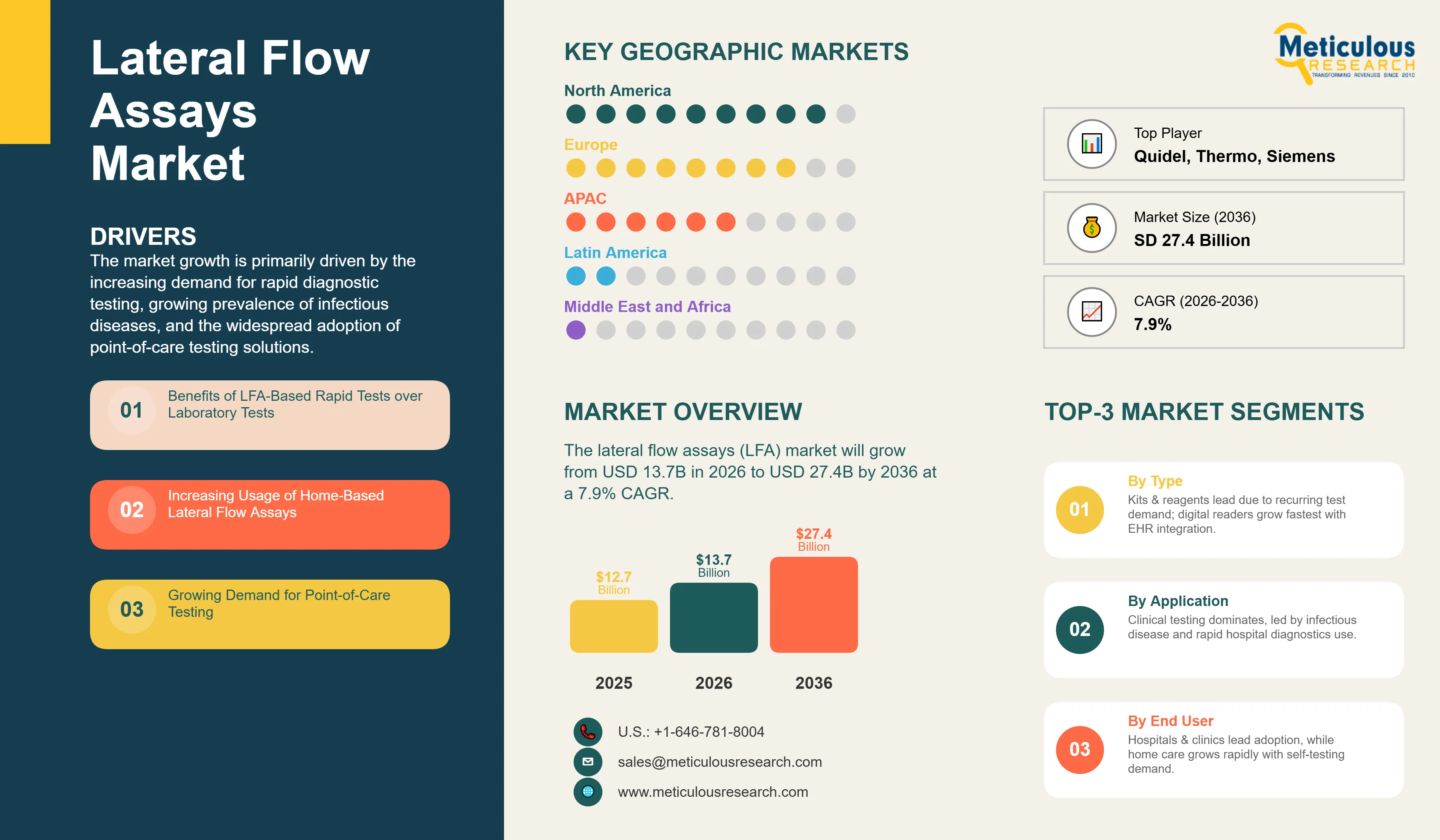
The global lateral flow assays (LFA) market is valued at USD 13.7 billion in 2026 and is expected to reach approximately USD 27.4 billion by 2036, at a CAGR of 7.9% from 2026 to 2036. The market growth is primarily driven by the increasing demand for rapid diagnostic testing, growing prevalence of infectious diseases, and the widespread adoption of point-of-care testing solutions. The advantages of lateral flow assays over traditional laboratory tests, including rapid turnaround time, ease of use, and cost-effectiveness, are fueling market expansion. Additionally, the post-pandemic emphasis on accessible diagnostics and the continuous innovations in LFA technology are expected to sustain market growth throughout the forecast period.
Market Highlights: Global Lateral Flow Assays Market
- The global lateral flow assays market is projected to reach USD 27.4 billion by 2036.
- The market is expected to grow at a CAGR of 7.9% from 2026 to 2036.
- Kits and reagents segment is expected to dominate the market by product type, in 2026.
- Digital and mobile readers are projected to witness the fastest growth among reader types from 2026 to 2036
- Clinical testing is expected to be the largest application segment, in 2026
- Infectious disease testing is projected to account for the largest share of clinical testing applications, in 2026
- Hospitals and clinics are expected to be the dominant end-user segment, in 2026
- North America is projected to hold the largest market share of the global lateral flow assays market, in 2026
Market Overview and Insights:
Click here to: Get Free Sample Pages of this Report
The global lateral flow assays market is experiencing significant growth, driven by the increasing demand for rapid, accessible diagnostic solutions across healthcare settings. Lateral flow assays have become indispensable tools for point-of-care testing, enabling healthcare professionals and consumers to obtain diagnostic results within minutes rather than hours or days required by conventional laboratory tests.
The market encompasses a diverse range of products, including rapid test kits for infectious diseases, pregnancy testing, cardiac markers, cholesterol screening, and drug abuse detection. The competitive landscape includes established diagnostic companies such as Abbott Laboratories, F. Hoffmann-La Roche AG, Siemens Healthineers AG, Becton Dickinson and Company, and Thermo Fisher Scientific, Inc., alongside emerging specialized manufacturers.
The expansion of lateral flow assay applications beyond traditional clinical diagnostics into veterinary diagnostics, food safety testing, and pharmaceutical quality control has broadened the addressable market. The integration of digital readers and mobile connectivity has enhanced the functionality and reliability of lateral flow assay results, enabling remote monitoring and data management capabilities. Furthermore, the growing emphasis on home-based testing and self-diagnostics has created new distribution channels and consumer segments for lateral flow assay products.
What are the Key Trends in the Lateral Flow Assays Market?
Integration of Digital and Mobile Reader Technologies
The integration of digital and mobile reader technologies is transforming the lateral flow assays market. Digital readers provide quantitative results, improving the accuracy and reliability of lateral flow assay testing compared to visual interpretation. Mobile-based readers enable connectivity with smartphones and other devices, facilitating data capture, storage, and transmission. This technological advancement is particularly valuable in remote and resource-limited settings where access to traditional laboratory infrastructure is limited. Companies such as Abbott Laboratories and Siemens Healthineers AG are investing in digital reader development to enhance the clinical utility of lateral flow assays. The adoption of digital readers is expected to expand the market beyond traditional point-of-care settings into home healthcare and telemedicine applications.
Expansion of Multiplex Detection and Multi-Analyte Testing
The development of multiplex lateral flow assays capable of detecting multiple analytes simultaneously is a significant market trend. Multiplex assays enable comprehensive diagnostic evaluation in a single test, improving efficiency and reducing testing costs. This advancement is particularly valuable for infectious disease screening, where simultaneous detection of multiple pathogens can guide treatment decisions. The technical challenges associated with developing reliable multiplex assays are being addressed through innovations in assay design, reagent formulation, and reader technology. Leading manufacturers are expanding their multiplex assay portfolios to address the growing demand for comprehensive diagnostic solutions. The market for multiplex lateral flow assays is projected to grow faster than single-analyte assays, reflecting the increasing preference for comprehensive diagnostic testing.
Market Summary
|
Parameters
|
Details
|
|
Market Size by 2036
|
USD 27.4 Billion
|
|
Market Size in 2026
|
USD 13.7 Billion
|
|
Revenue Growth Rate (2026-2036)
|
CAGR of 7.9%
|
|
Dominating Product Type
|
Kits & Reagents
|
|
Fastest Growing Reader Type
|
Digital/Mobile Readers
|
|
Largest Application Segment
|
Clinical Testing
|
|
Dominant Clinical Testing Type
|
Infectious Disease Testing
|
|
Largest End-User Segment
|
Hospitals & Clinics
|
|
Leading Geographic Region
|
North America
|
|
Base Year
|
2025
|
|
Forecast Period
|
2026 to 2036
|
Market Dynamics
Drivers: Benefits of LFA-Based Rapid Tests and Growing Demand for Point-of-Care Testing
The primary drivers of the lateral flow assays market include the significant advantages of LFA-based rapid tests over conventional laboratory tests. Lateral flow assays offer rapid turnaround times, ease of use, cost-effectiveness, and minimal equipment requirements, making them ideal for point-of-care testing in diverse settings. The growing prevalence of chronic and infectious diseases, coupled with an aging global population, is increasing the demand for accessible diagnostic solutions. Additionally, the post-COVID-19 emphasis on home-based testing and self-diagnostics has expanded the market beyond traditional healthcare settings. Government initiatives promoting health awareness and the adoption of preventive healthcare measures are further supporting market growth.
Opportunity: Evolving Applications and Emerging Markets
The expanding applications of lateral flow assays beyond infectious disease testing present significant growth opportunities. The adoption of lateral flow assays in veterinary diagnostics, food safety testing, and pharmaceutical quality control is broadening the addressable market. Emerging economies in Asia-Pacific, Latin America, and the Middle East & Africa are experiencing rapid growth in demand for accessible diagnostic solutions, driven by improving healthcare infrastructure and increasing healthcare spending. The development of novel lateral flow assay formats and the integration of advanced technologies such as artificial intelligence and machine learning are expected to create new market opportunities. Furthermore, the growing emphasis on personalized medicine and the development of assays for emerging biomarkers are expected to drive market expansion.
Product Type Insights
Why Do Kits & Reagents Dominate the Market?
Kits and reagents represent the largest product segment in the lateral flow assays market, in 2026. The dominance of this segment is attributed to the high volume of diagnostic tests performed globally and the recurring nature of kit and reagent purchases. Healthcare facilities, diagnostic laboratories, and home users require continuous supplies of lateral flow assay kits for ongoing diagnostic testing. The cost-effectiveness of lateral flow assay kits compared to alternative diagnostic methods has driven their widespread adoption. Established manufacturers such as Abbott Laboratories and F. Hoffmann-La Roche AG have built substantial market presence through their comprehensive kit and reagent portfolios.
How Are Digital Readers Transforming the Market?
Digital and mobile lateral flow readers are the fastest-growing product segment. These readers provide quantitative results, eliminating the subjectivity associated with visual interpretation of lateral flow assays. The integration of connectivity features enables data transmission to electronic health records and laboratory information systems, improving workflow efficiency. Digital readers are particularly valuable in clinical laboratories and high-volume testing environments. The development of portable and user-friendly digital readers is expanding their adoption in point-of-care settings and home healthcare applications.
Application Insights
Why Does Clinical Testing Lead the Market?
Clinical testing is the largest application segment for lateral flow assays, driven by the widespread use of rapid diagnostic tests in healthcare settings. Infectious disease testing, including tests for COVID-19, influenza, and other respiratory pathogens, represents the largest sub-segment within clinical testing. The rapid turnaround time and ease of use of lateral flow assays make them ideal for screening and diagnostic purposes in hospitals, clinics, and emergency departments. Pregnancy and fertility testing, cardiac marker testing, and cholesterol screening are additional clinical applications driving market growth. The post-pandemic increase in demand for rapid diagnostic testing has significantly expanded the clinical testing segment.
What Are the Emerging Applications?
Emerging applications of lateral flow assays in veterinary diagnostics, food safety testing, and drug development are creating new market opportunities. In veterinary medicine, lateral flow assays are used for rapid diagnosis of infectious diseases in animals, supporting animal health management and food security. In food safety, lateral flow assays enable rapid detection of pathogens and contaminants, supporting food quality assurance and regulatory compliance. In pharmaceutical development, lateral flow assays are used for quality control and biomarker detection, supporting drug development processes.
End-User Insights
How Do Hospitals & Clinics Dominate the Market?
Hospitals and clinics hold the largest share of the overall lateral flow assays, driven by the high volume of diagnostic testing performed in these settings. The rapid turnaround time and ease of use of lateral flow assays make them essential tools for emergency departments, outpatient clinics, and inpatient units. The integration of lateral flow assays into clinical workflows has improved patient care by enabling rapid diagnosis and treatment decisions. The adoption of digital readers in hospitals has further enhanced the utility of lateral flow assays by providing quantitative results and enabling data integration with electronic health records.
How Is Home Care Growing Rapidly?
Home care settings are experiencing rapid growth in lateral flow assay adoption, driven by the increasing emphasis on home-based testing and self-diagnostics. The ease of use and minimal equipment requirements of lateral flow assays make them suitable for home testing applications. The post-COVID-19 expansion of home healthcare services and the growing consumer demand for convenient diagnostic solutions are driving the growth of the home care segment. Manufacturers are developing user-friendly lateral flow assay kits specifically designed for home use, with simplified instructions and clear result interpretation.
Regional Insights
How Does North America Maintain Its Leadership?
North America is expected to hold the largest market share for lateral flow assays, driven by advanced healthcare infrastructure, high healthcare spending, and strong regulatory frameworks. The United States has been at the forefront of lateral flow assay innovation and adoption, with significant investments in diagnostic technology development. The widespread adoption of point-of-care testing in U.S. healthcare settings and the strong presence of major manufacturers such as Abbott Laboratories and Becton Dickinson and Company have solidified market leadership of this region. The post-pandemic emphasis on rapid diagnostic testing has further accelerated market growth in the region.
Which Regions Are Experiencing Rapid Growth?
Europe is projected to experience steady growth in the lateral flow assays market, driven by supportive regulatory frameworks and the adoption of point-of-care testing in healthcare systems. Asia-Pacific is emerging as the fastest-growing region, with increasing healthcare spending, improving healthcare infrastructure, and growing awareness of diagnostic testing in countries such as China, India, and Japan. The expansion of diagnostic capabilities in emerging economies and the growing demand for accessible healthcare solutions are driving rapid market growth in Asia-Pacific and other emerging regions.
Key Players in the Global Lateral Flow Assays Market
The key players profiled in the global lateral flow assays market report include Abbott Laboratories, F. Hoffmann-La Roche AG, Becton Dickinson and Company, Siemens Healthineers AG, Thermo Fisher Scientific, Inc., Bio-Rad Laboratories, Inc., Biomérieux S.A., QIAGEN N.V., PerkinElmer, Inc., Quidel Corporation, and Merck KGAA.
Key Questions Answered