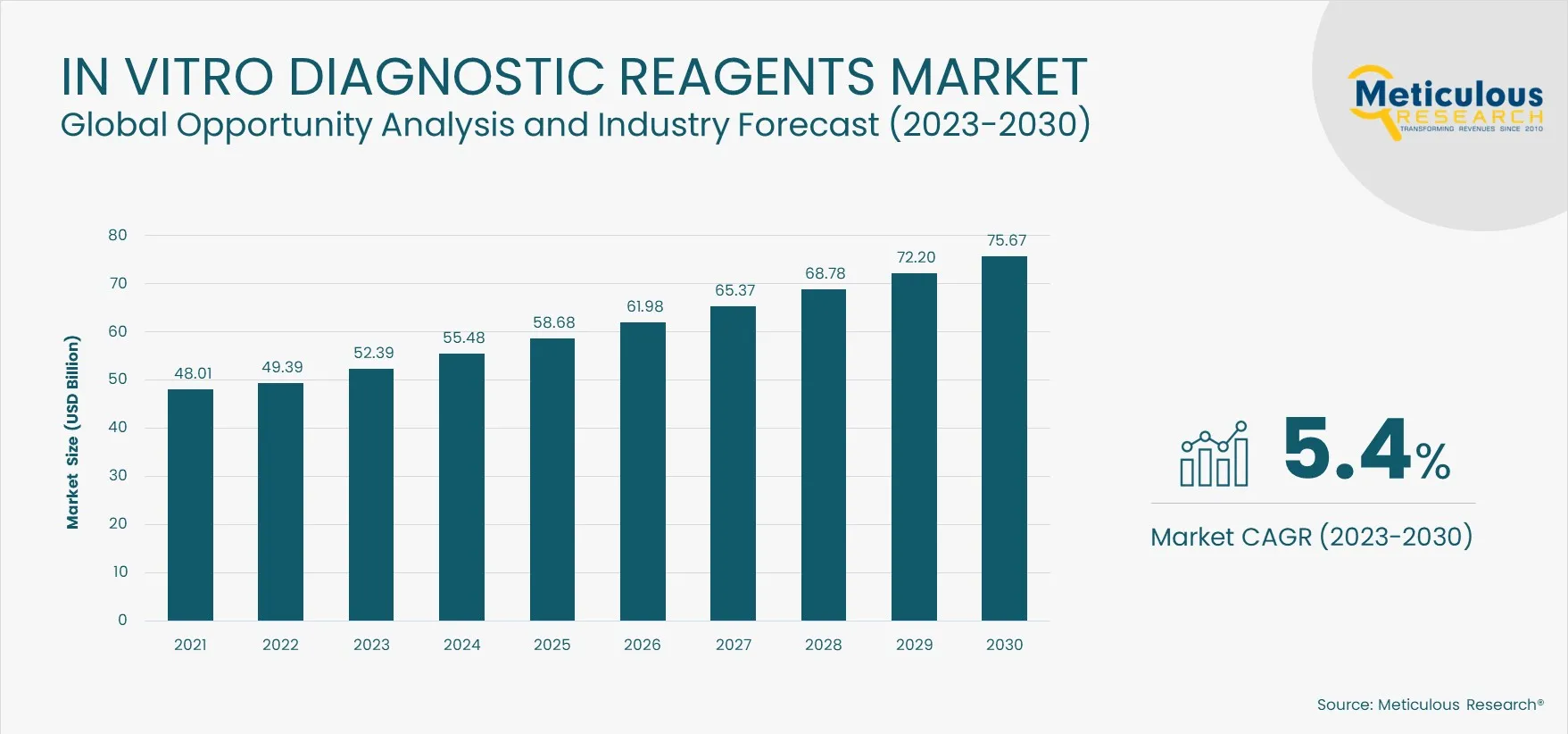
The In Vitro Diagnostic Reagents Market is projected to reach $75.67 billion by 2030, at a CAGR of 5.4% from 2023 to 2030. IVD reagents are utilized in laboratory tests to identify the absence or presence of a certain condition in biological samples such as urine, blood, and other bodily fluids. IVD reagents include antibodies, oligonucleotides, nucleic acid probes, and other biomolecules specifically developed to interact with a specific biomarker or target molecule linked to a condition or disease. IVD reagents are used in various diagnostic procedures, including immunoassay/immunochemistry, molecular diagnostics, microbiology tests, and others, to diagnose infectious and chronic disorders.
The growth of this market is driven by the increasing prevalence of infectious diseases, increasing funding for research activities, the rising adoption of advanced diagnostic technologies, the growth in proteomics and genomics research studies, and the growing number of clinical laboratories offering advanced diagnostic testing menus. Additionally, emerging economies and the growing demand for protein therapeutics and personalized medicines are expected to offer significant market growth opportunities.
However, the stringent regulations for antigen-specific reagents restrain the growth of this market. Additionally, the high costs of reagents and the unfavorable reimbursement scenario pose a significant challenge to the market's growth.
Increasing Prevalence of Infectious Diseases is Driving the IVD Reagents Market’s Growth
Infectious diseases are mainly caused by organisms such as bacteria, viruses, fungi, and parasites. Some infectious diseases get transmitted from person to person or through bites from insects or animals. The burden of infectious diseases has increased globally, driving the demand for safe and effective diagnostic assays. Hence, the demand for IVD reagents is expected to grow with the increased utilization of diagnostic assays. The following statistics indicate the increased prevalence of infectious diseases:
-
In 2021, 39 million people had HIV globally, of which 1.5 million were children. There were 20.8 million (53%) people with HIV in eastern and southern Africa, 5 million (13%) in western and central Africa, 6.5 million (15%) in Asia and the Pacific, and 2.3 million (5%) in Western and Central Europe and North America.
- According to an article published in the United Nations digital magazine Africa Renewal in July 2022, the risk of outbreaks caused by zoonotic pathogens such as the Ebola & monkeypox viruses that originate in animals and infect humans is rising. There has been a 63.0% increase in the number of zoonotic disease outbreaks in Africa from 2012–2022 compared to 2001– 2011 (Source: WHO).
- According to the WHO, about 1.6 million people died from tuberculosis in 2021. The disease was the 13th leading cause of death globally. In 2021, there were 10.6 million new cases of tuberculosis, of which 3.4 million were women and 1.2 million were children. The burden of drug-resistant TB (DR-TB) increased by 3% between 2020 and 2021, with 450,000 new cases of rifampicin-resistant TB (RR-TB) recorded in 2021.
Thus, the increasing prevalence of infectious diseases is expected to boost the need for diagnosis, driving the demand for IVD reagents.
Growing Demand for Protein Therapeutics and Personalized Medicines to Offer Market Growth Opportunities
Protein therapeutics is a branch of medicine that can serve patients needing novel therapies. Advances in protein-engineering technologies (such as mass spectrometry), molecular genetics, and production technologies are key drivers of protein therapeutic practices. Drug developers are exploiting the functional characteristics of desired proteins to maintain and enhance product safety or efficacy. The fastest-growing class of therapeutic proteins is an antibody that includes bispecific mAbs, multispecific fusion proteins, and mAbs conjugated with small-molecule drugs designed to treat cancers, immune disorders, infections, and other diseases.
According to a research article published in F1000Research, recent advances in protein purification technologies and expression systems have increased the production of therapeutic peptides while maintaining product safety or efficacy or both at the same time. Further, government initiatives supporting the development of personalized medicines (which largely include monoclonal antibodies) have boosted antibody production.
In March 2020, scientists in South Africa developed a new antibody technology for precision medicine. A group of researchers from the University of Cape Town’s Medical Biotechnology and Immunotherapy Research Unit published a paper explaining how certain proteins based on antibodies, called SNAP-tag fusion proteins, may help reduce the cost and time needed to develop new personalized therapies. The fact that antibodies have these specific targets is important in developing targeted therapies. Scientists can generate new protein combinations for various applications by rearranging genetic material such as antibody genes. An example of precision medicine is using antibody technologies to identify breast cancer patients treated with trastuzumab.
Thus, the increasing demand for therapeutic proteins and personalized medicines and the increase in approvals are expected to create growth opportunities for the players operating in the IVD reagents market.
Key Findings in the IVD Reagents Market Study:
Among the Types, the Antibodies Segment is Expected to Account for the Largest Share of the Market in 2023
Among the types, in 2023, the antibodies segment is expected to account for the largest share of the IVD reagents market. Advantages offered by the segment, like the inherent specificity, high throughput, high sensitivity, and low cost, drive the growth of this technology segment. Further, the increasing use of immunoassays in POC & infectious disease testing, the development of novel tests, and the rising demand for immunoassay-based tests are the factors contributing to the largest share of this segment.
-Reagents-Market.webp)
Click here to: Get Free Sample Pages of this Report
Among the Technologies, the Immunoassay/Immunochemistry Segment is Expected to Account for the Largest Share of the Market in 2023
Among the technologies, in 2023, the immunoassay/immunochemistry segment is expected to account for the largest share of the IVD reagents market. The increasing use of immunoassays in POC and infectious disease testing, the development of novel tests, the increasing usage of miniaturized devices, and the rising demand for immunoassay-based tests are the factors contributing to the largest share of this segment.
Among the Uses, the Clinical Use Segment is Expected to Account for the Largest Share of the Market in 2023
Among the uses, in 2023, the clinical use segment is expected to account for the largest share of the IVD reagents market. Initiatives by manufacturers for developing advanced IVD reagents for clinical applications, ongoing clinical trials, and exemption from the investigational device (IDE) requirements are the factors contributing to the largest share of this segment.
Among the end users, the IVD Manufacturers Segment is Expected to Account for the Largest share of the Market in 2023
Among the end users, in 2023, the IVD manufacturers segment is expected to account for the largest share of the IVD reagents market. The increasing demand for innovative products, growing incidence of chronic diseases, and increasing demand for pharmaceutical & biopharmaceutical products from emerging countries are the factors contributing to the largest share of this segment.
Asia-Pacific: Fastest-growing Regional Market
The growth of this regional market is attributed to rapid urbanization, increasing investments by healthcare providers towards infrastructure improvement, the presence of key IVD reagents companies in countries like China, India, Singapore, and South Korea, and government investments in research and development activities. Furthermore, the need to manage the growing burden of infectious diseases, growing accessibility to healthcare services & advanced products, growing income levels of the middle-class population, and increasing number of hospitals and clinics contribute to the growth of this market.
Key Players
The report offers a competitive landscape based on an extensive assessment of the product offerings and geographic presence of leading market players and the key growth strategies adopted by them in the last three to four years. The key players profiled in the IVD reagents market report are Thermo Fisher Scientific (U.S.), Becton, Dickinson and Company (U.S.), Merck KGaA (Germany), Bio-Rad Laboratories, Inc. (U.S.), Agilent Technologies, Inc. (U.S.), Abcam plc (U.K.), Hologic Inc. (U.S.), InBios International, Inc. (U.S.), SDIX, LLC (U.S.), BioTechne Corporation (U.S.), Bio-Synthesis Inc. (U.S.), RayBiotech, Inc. (U.S.), Sino Biological Inc. (China), Oy Medix Biochemica Ab (Finland), QIAGEN N.V. (Netherlands), and Beckman Coulter, Inc. (U.S.).
Scope of the Report:
IVD Reagents Market Size & Trend Analysis, by Type
- Antibodies
- Antibodies Market, By Type
- Monoclonal Antibodies
- Polyclonal Antibodies
- Antibodies Market, By Target
- Cluster of Differentiation (CD)
- T-cell Receptor (TCR)
- Epidermal Growth Factor Receptor (EGFR)
- Other Targets
- Purified Proteins and Peptides
- Oligonucleotides
- Nucleic Acid Probes
- Other Reagents
(Notes: 1. Other targets include Nucleic BCMA, DLL/Notch, EpCAM, HER2, PDGFRα, Claudin 18.2, SLAMF7, and DKK1.
B-cell maturation antigen (BCMA).
2. Other reagents include blocking agents, detergents, surfactants, wetting agents, buffers, stabilizers, purified water, dyes,
standards and controls, enzymes, cell culture reagents, and anti-coagulants.)
IVD Reagents Market Size & Trend Analysis, by Technology
- Immunoassay/Immunochemistry
- Enzyme-linked Immunosorbent Assays (ELISA) and Enzyme-linked Immunospot Assays (ELISpot)
- Lateral Flow Assays (LFA)/Rapid Tests
- Western Blotting (WB)
- Immunofluorescence (IF)
- Functional Assays
- Immunoprecipitation (IP)
- Other Immunoassay Technologies
- Biochemistry/Clinical Chemistry
- Molecular Diagnostics
- Polymerase Chain Reaction (PCR)
- Hybridization
- Isothermal Nucleic Acid Amplification Technology
- DNA Sequencing & Next-generation Sequencing
- Microarray
- Other Molecular Diagnostic Technologies
- Microbiology
- Hematology
- Coagulation/Hemostasis
- Urinalysis
- Other IVD Technologies
(Note: 1. Other technologies include virus neutralization tests, agar gel immunodiffusion, and radioimmunoassay.
2. Other molecular diagnostic technologies include electrophoresis, ligase chain reaction (LCR), northern blot, and southern blot.
3. Other IVD technologies include anatomical pathology, histochemistry, and whole blood glucose monitoring.)
IVD Reagents Market Size & Trend Analysis, by Use
- Clinical Use
- Research Use Only (RUO)
- Analyte-specific Reagents (ASR)
IVD Reagents Market Size & Trend Analysis, by End User
- Reference Laboratories
- IVD Manufacturers
- Academic Institutes and Research Laboratories
- Hospital Laboratories
IVD Reagents Market Size & Trend Analysis, by Geography
- North America
- Europe
- Germany
- France
- Italy
- U.K.
- Spain
- Switzerland
- Netherlands
- Rest of Europe
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Singapore
- Rest of Asia-Pacific
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Middle East & Africa
Key questions answered in the report:

-Reagents-Market.webp)