Resources
About Us
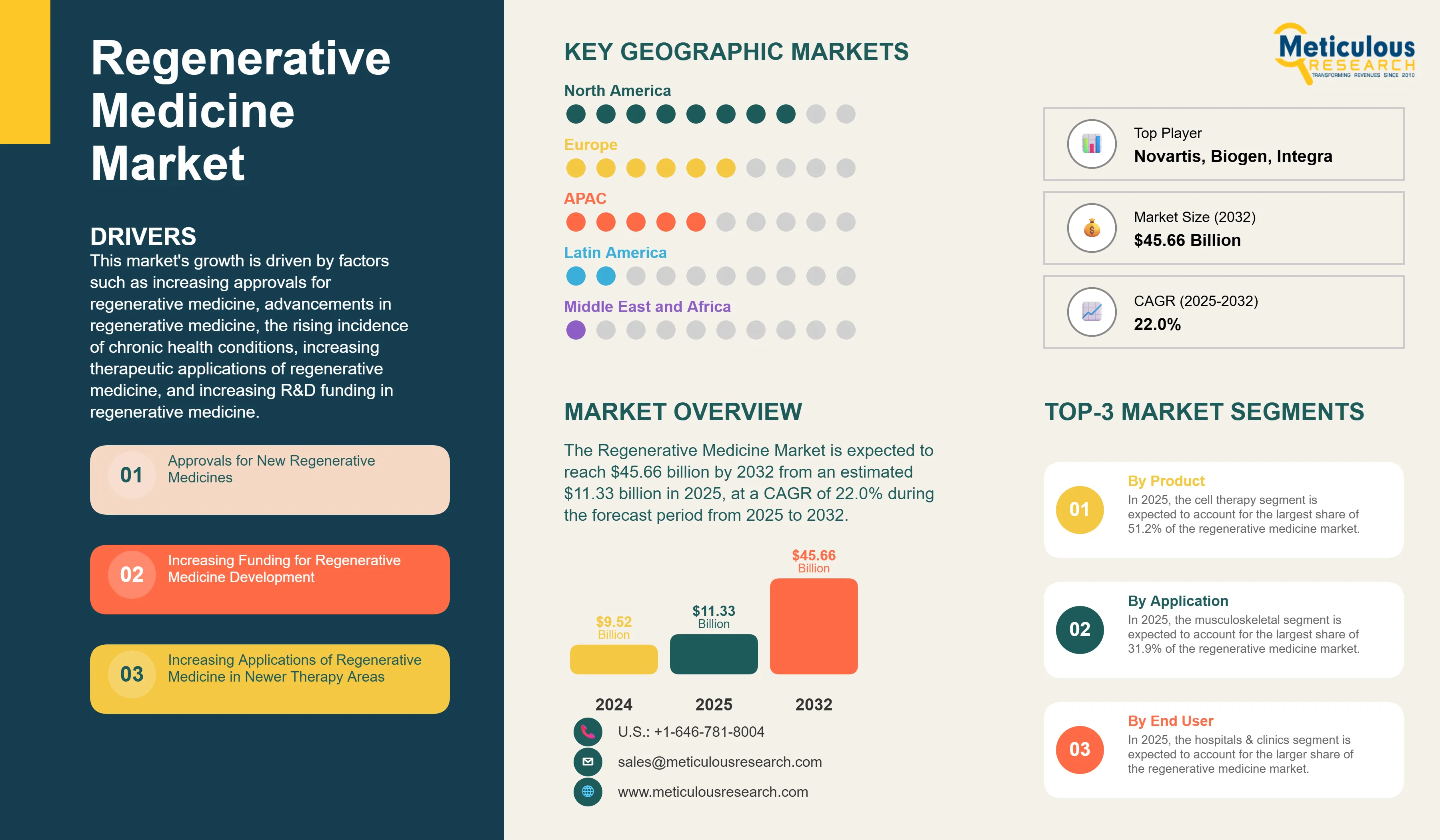
Regenerative Medicine Market Size, Share, Forecast, & Trends Analysis by Product (Gene Therapy, Stem Cell, Tissue Engineering) Application (Musculoskeletal, Cancer, Cardiovascular, Immunology, Dermatology, Ophthalmology) End User - Global Forecast to 2032
Report ID: MRHC - 1041181 Pages: 240 Jun-2025 Formats*: PDF Category: Healthcare Delivery: 2 to 4 Hours Download Free Sample ReportThis market's growth is driven by factors such as increasing approvals for regenerative medicine, advancements in regenerative medicine, the rising incidence of chronic health conditions, increasing therapeutic applications of regenerative medicine, and increasing R&D funding in regenerative medicine.
Furthermore, the increasing demand for personalized medicine, the growing need for healthy organs for transplant, and the growing number of drugs in the pipeline are expected to create market growth opportunities.
Regenerative medicine harnesses the body’s inherent healing capabilities to restore, repair, or cultivate damaged or diseased tissues. The regenerative medicine field has witnessed a surge in interest in recent years, driven by its promising applications in oncology, cardiology, neurology, and other medical domains. With the increasing application areas of regenerative medicine, regulatory bodies are approving the use of regenerative medicine for treating different diseases. Some instances of such approvals are as follows:
Regenerative medicine is proving to be an effective therapy for various medical conditions, including cardiovascular diseases, ophthalmic diseases, oncology, neurological diseases, immunology & inflammation, musculoskeletal diseases, and dermatological diseases. The growing incidence of these target diseases is a key driver for market growth.
Neurodegenerative disorders, characterized by neuron loss, represent a significant portion of these conditions. Major neurodegenerative disorders include Alzheimer's disease and other memory disorders, Parkinson's disease, Ataxia, and Huntington's disease.
According to the Alzheimer's Association (U.S.), the number of people living with Alzheimer's disease is expected to reach 13 million by 2050 from 6.9 million in 2024 in the U.S. The care cost for people living with dementia is estimated to reach about $1 trillion in 2050 in the U.S. To reduce the high prevalence and cost associated with the disease, healthcare providers are focusing on the use of stem cell therapy for therapeutic purposes. For instance, in April 2024, Regeneration Biomedical (U.S.) started the human clinical trial for RB-ADSC, an autologous stem cell treatment for people living with mild-to-moderate Alzheimer’s disease.
The global demand for organ transplantation is steadily rising due to various factors, such as increasing cases of chronic diseases leading to organ damage, genetic disorders, accidental injuries, and physical trauma to organs. According to the U.S. Health Resources & Services Administration, as of March 2024, there were 106,441 patients in the U.S. awaiting organ donation.
Regenerative medicine modalities, such as stem cell therapy and tissue engineering, play a crucial role in repairing or regenerating damaged organs, thereby reducing reliance on donor organs and enhancing transplant outcomes. While the development of organs in vitro is still in its nascent stage, regenerative medicine presents an opportunity for organ development using a patient's cells, thereby reducing the likelihood of rejection and the need for donor organs.
3D bioprinting is a cutting-edge technique in regenerative medicine, utilizing biomaterials and cells, as opposed to traditional polymers and metals, to fabricate functional 3D tissues. This technology has gained prominence for creating highly specialized tissue models, complementing conventional tissue engineering methods.
In tissue engineering, 3D bioprinting plays a crucial role in assembling living cells, biochemicals, or biomaterials into functional tissue-like structures. 3D bioprinting enables the integration of engineered tissue with natural tissue, facilitating the restoration of organ and tissue function.
Due to its advantages, 3D bioprinting is gradually replacing traditional technologies. Unlike conventional methods, 3D bioprinting ensures accurate scaffold creation and proper cell line interaction. Moreover, it enables the fabrication of tissue components from patients' stem cells, thereby reducing the risk of rejection and graft-versus-host reactions, leading to increased adoption of this innovative approach.
Induced Pluripotent Stem (iPS) cells are taken from a patient and reprogrammed so that they can differentiate into any type of cell in the body. By maintaining the patient's genetic code, iPS cells play a crucial role in disease modeling and regenerative medicine.
Induced Pluripotent Stem Cells (iPSC) are derived from skin or blood cells that have been reprogrammed back into an embryonic-like pluripotent state that enables the development of an unlimited source of any type of human cell needed for therapeutic purposes. For example, iPSCs can be manipulated into becoming beta islet cells to treat diabetes, blood cells to create new cancer-free blood for leukemia patients and neurons to treat neurological disorders. As research on induced pluripotent stem cells advances, iPSCs are expected to emerge as a promising solution in regenerative medicine.
Based on product, the regenerative medicine market is segmented into gene therapy, cell therapy, and tissue engineering products. In 2024, the cell therapy segment is expected to account for the largest share of 51.2% of the regenerative medicine market. This segment is projected to reach $23,234.7 million by 2031 from an estimated $4,870.8 million in 2024, at a CAGR of 25.0% during the forecast period. This segment’s large market share is attributed to the expanding use of cell therapies in treating various conditions such as cancer, autoimmune diseases, cardiovascular ailments, and other chronic illnesses.
The rising prevalence of long-term conditions has led to a surge in the adoption of cell therapies. According to the Centers for Disease Control and Prevention, 6 out of 10 adults in the U.S. have at least one chronic condition, with 4 in 10 adults affected by two or more. In cell therapy, damaged tissue is replaced with healthy counterparts or by stimulating tissue repair and regeneration. The segment's growth is further propelled by increased approvals for cell therapy and a growing body of positive clinical outcomes associated with regenerative cells.
Based on application, the regenerative medicine market is segmented into cardiology, ophthalmology, oncology, neurology, immunology & inflammation, musculoskeletal, dermatology, and other applications. In 2024, the musculoskeletal segment is expected to account for the largest share of 31.9% of the regenerative medicine market. Conditions that affect the joints and certain connective tissues are known as musculoskeletal conditions. The high incidence of bone-related injuries and the rising prevalence of musculoskeletal disease are increasing the demand for regenerative medicine.
According to the Australian Institute of Health and Welfare, around 7.3 million people (29%) in Australia are estimated to be living with chronic musculoskeletal conditions. Additionally, the increasing preference for regenerative medicine over surgery for muscle strain injuries in athletes and partial tendon tears is further contributing to the significant market share of this segment.
Based on end user, the regenerative medicine market is segmented into hospitals & clinics and ambulatory surgical centers. In 2024, the hospitals & clinics segment is expected to account for the larger share of the regenerative medicine market. This segment’s large market share can be attributed to the rising number of patient visits to hospitals, the growing number of hospitals, and the availability of advanced technological resources within these healthcare facilities. Additionally, funding is often provided to hospitals & clinics to establish dedicated regenerative medicine facilities. For instance, in October 2022, California's stem cell agency granted $8 million to Cedars-Sinai (U.S.), a non-profit teaching hospital and multi-specialty academic health science center. This grant aims to establish an Alpha Stem Cell Clinic, enhancing patient access to stem cell therapy.
In 2024, North America is expected to account for the largest share of the 41.8% of regenerative medicine market. North America’s significant market share can be attributed to the increasing prevalence of target diseases such as cancer and cardiovascular diseases, growing funding for the development of regenerative medicine, the presence of key market players in the region, and the rising number of clinical trials. According to the Alliance for Regenerative Medicine (U.S.), an international organization for regenerative medicine, North America witnessed a notable increase in the number of clinical trials, reaching 394 trials during the third quarter of 2023 from 339 trials in the fourth quarter of 2022. Similarly, the number of developers engaged in cell therapy manufacturing in North America increased from 272 to 435 during the same period. This surge in clinical trials and the involvement of key players in the development of regenerative medicine contribute to North America’s significant market share.
However, the market in Asia-Pacific is projected to register the highest CAGR of 25.0% during the forecast period. The growth of this regional market is attributed to the rising prevalence of target diseases, the growing adoption of stem cell technology, supportive government policies promoting the development of regenerative medicine, and increased funding and research and development (R&D) initiatives in this field. According to the Alliance for Regenerative Medicine (U.S.), investment in regenerative medicine in the Asia-Pacific region surged to $2.1 billion in the fourth quarter of 2023, up from $0.9 billion in the third quarter of the same year.
The report offers a competitive landscape based on an extensive assessment of the offerings and geographic presence of leading market players and the key growth strategies adopted by them over the past three to four years. The key players operating in the regenerative medicine market are Novartis AG (Switzerland), Biogen Inc. (U.S.), Kite Pharma, Inc. (U.S.), Spark Therapeutics, Inc. (U.S.), Integra LifeSciences Corporation (U.S.), Sarepta Therapeutics, Inc. (U.S.), Takeda Pharmaceutical Company Limited (Japan), Amgen Inc. (U.S.), CORESTEMCHEMON Inc. (South Korea), Smith & Nephew plc (England), Vertex Pharmaceuticals Incorporated (U.S.), CSL Behring, LLC (U.S.), Janssen Global Services, LLC (U.S.), Medtronic plc (Ireland), AbbVie Inc. (U.S.), Bristol-Myers Squibb Company (U.S.), Ferring Pharmaceuticals A/S (Sweden), Pfizer Inc. (U.S.), bluebird bio Inc. (U.S.), and Vericel Corporation (U.S.).
In May 2024, Bristol-Myers Squibb Company (U.S.) received approval from the U.S. FDA for Breyanzi, a CAR T-cell therapy used to treat relapsed or refractory mantle cell lymphoma (MCL) and refractory follicular lymphoma in adult patients.
|
Particulars |
Details |
|
Number of Pages |
240 |
|
Format |
|
|
Forecast Period |
2025-2032 |
|
Base Year |
2024 |
|
CAGR |
22% |
|
Estimated Market Size (Value) |
$45.66 billion by 2032 |
|
Segments Covered |
By Product
By Application
By End User
|
|
Countries Covered |
North America (U.S. and Canada), Europe (Germany, France, U.K., Italy, Spain, Netherlands, Sweden, Switzerland, and Rest of Europe), Asia-Pacific (China, Japan, India, South Korea, Australia, and Rest of Asia-Pacific), Latin America (Brazil, Mexico, Rest of Latin America), and Middle East & Africa |
|
Key Companies |
Novartis AG (Switzerland), Biogen Inc. (U.S.), Kite Pharma, Inc. (U.S.), Spark Therapeutics, Inc. (U.S.), Integra LifeSciences Corporation (U.S.), Sarepta Therapeutics, Inc. (U.S.), Takeda Pharmaceutical Company Limited (Japan), Amgen Inc. (U.S.), CORESTEMCHEMON Inc. (South Korea), Smith & Nephew plc (England), Vertex Pharmaceuticals Incorporated (U.S.), CSL Behring, LLC (U.S.), Janssen Global Services, LLC (U.S.), Medtronic plc (Ireland), AbbVie Inc. (U.S.), Bristol-Myers Squibb Company (U.S.), Ferring Pharmaceuticals A/S (Sweden), Pfizer Inc. (U.S.), bluebird bio Inc. (U.S.), and Vericel Corporation (U.S.). |
The regenerative medicine market report analyzes various segments of regenerative medicine, such as product, application, and end user, at the regional and country levels. It also provides insights on factors impacting market growth, regulatory analysis, investment and funding scenarios, pipeline analysis, and Porter’s five forces analysis.
The Regenerative Medicine Market was valued at $9.52 billion in 2024. This market is expected to reach $45.66 billion by 2032 from an estimated $11.33 billion in 2025, at a CAGR of 22.0% during the forecast period from 2025 to 2032.
Among the products covered in this report, in 2024, the cell therapy segment is expected to account for the largest share of 51.2% of the regenerative medicine market. Stem cell therapy is a minimally invasive procedure, reducing the need for extensive surgeries and lengthy recovery periods. Since it utilizes the patient's body cells, it minimizes the risk of side effects and provides personalized treatment, making it a preferred option over other therapies.
The growth of this market is driven by factors such as the growing drug approvals for regenerative medicine, advancements in regenerative medicine, the rising incidences of target diseases, the expanding usage of regenerative medicine in newer therapies, and increasing funding for research & development of regenerative medicine. Furthermore, the growing demand for personalized medicine, the soaring demand for organ transplants, and the rising number of drugs in the pipeline are expected to create market growth opportunities.
The key players operating in the regenerative medicine market are Novartis AG (Switzerland), Biogen Inc. (U.S.), Kite Pharma, Inc. (U.S.), Spark Therapeutics, Inc. (U.S.), Integra LifeSciences Corporation (U.S.), Sarepta Therapeutics, Inc. (U.S.), Takeda Pharmaceutical Company Limited (Japan), Amgen Inc. (U.S.), CORESTEMCHEMON Inc. (South Korea), Smith & Nephew plc (England), Vertex Pharmaceuticals Incorporated (U.S.), CSL Behring, LLC (U.S.), Janssen Global Services, LLC (U.S.), Medtronic plc (Ireland), AbbVie Inc. (U.S.), Bristol-Myers Squibb Company (U.S.), Ferring Pharmaceuticals A/S (Sweden), Pfizer Inc. (U.S.), bluebird bio Inc. (U.S.), and Vericel Corporation (U.S.).
Asia-Pacific is slated to register the highest growth rate of 25.0% during the forecast period. This regional market's growth is attributed to the rising prevalence of target diseases, the growing adoption of stem cell technology, supportive government policies promoting the development of regenerative medicine, and increased funding and research and development (R&D) initiatives in this field.
























Published Date: Jan-2025
Published Date: Jul-2023
Please enter your corporate email id here to view sample report.
Subscribe to get the latest industry updates